- For U.S. Healthcare Professionals Only
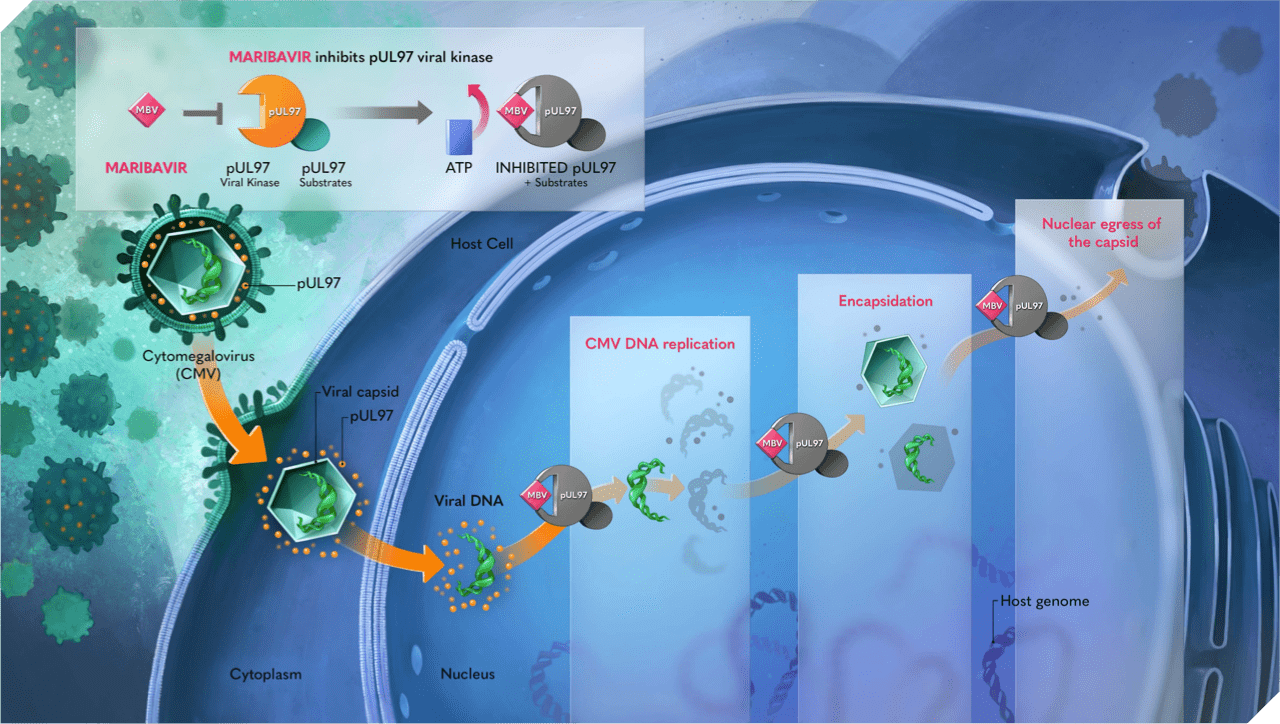
Mechanism of Action1,5-10
- Maribavir is an antiviral drug against human CMV1
- Its action is mediated by competitive inhibition of the protein kinase activity of human CMV enzyme pUL97 and its natural substrates,* which results in inhibition of the phosphorylation of proteins.1,5,6 This results in the downstream inhibition of:
- CMV DNA replication7,8
- Viral encapsidation8
- Nuclear egress of viral capsids9
*The natural substrates (phosphorylation targets) of pUL97 include ppUL44, pp65, RNA polymerase II large subunit, retinoblastoma protein, lamin A and C, p32, nuclear egress complex (UL50, UL53), histone deacetylase 1, and eukaryotic elongation factor delta.6,10

You are being directed to another website.
